|
As the first ionisation energy decreases as you move down the group, less energy is required to move the electrons, therefore the elements become more reactive. Reactions of Group 2 elements consist of the elements losing electrons to form ions with a +2 charge. 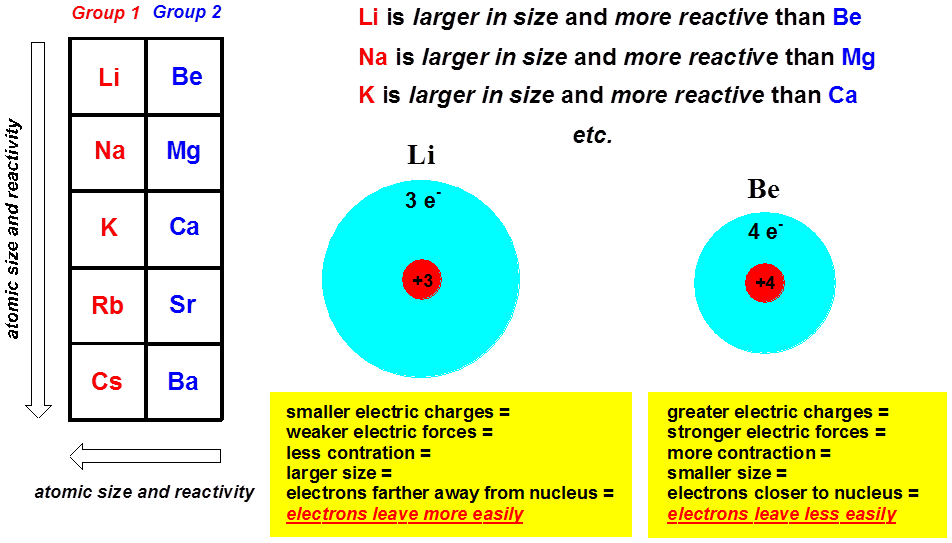
As you move down Group 2, the reactivity of the elements increases.When they react with water, they form the hydroxide and hydrogen gas. The attraction between the outermost electrons and the nucleus decreases, therefore less energy is required to remove them. They exist almost exclusively in the +2 oxidation state. The shielding effect on the outermost electrons also increases due to the presence of more inner electron shells containing electrons. More electron shells are added as you move down Group 2 which means the atomic radius increases and therefore the outermost shell becomes further and further away from the positive nucleus. As you move down Group 2, the first ionisation energy of the elements decreases.Between beryllium and radium, there is a change in the melting point trend at magnesium as it has a different crystalline structure.įirst Ionisation Energy Trend Down Group 2 The arrangement of the Group 2 ions in the metal affects the melting point.Less energy is needed to break the metallic bonds as the delocalised electrons are further away from the positive nuclei because of an increased ionic radius, therefore there is a decreased attraction between them. The charge of +2 of of the positive ions does not change either. As you move down the group, the radius of the metal ions increases but the number of delocalised electrons stays the same per atom. Group 2 elements generally have metallic bonding as they are metallic structures. As you move down Group 2, the melting points of the elements decreases.The atomic radius of Group 2 elements decreases because more and more electron shells are being added down the group. This trend is consistent with the expected reactivity based on ionic radii. As you move down Group 2, the atomic radius of the elements increases.Trends in Group 2 Elements Atomic Radius Trend Down Group 2 Elements in group 2 will lose two electrons to gain a full outer shell of electrons, to form ions with a +2 charge when they react. The outer shell of group 2 elements contains two electrons and is therefore represented by the sub shell s2. All the alkaline earth metals are highly reactive elements since they have a strong tendency to lose the two valences s-electrons to form the corresponding. 
So, it becomes comparatively easier to extract such least reactive metals. For example, Gold, Platinum is found in free state. Some of these metals are found in the earth’s crust in their free state. Group 2 elements are collectively known as the Alkaline Earth Metals. Reactivity Series - Reactivity Series of Metals refers to the Arrangement of Metals in the Descending Order of their Reactivities. Group 2, The Alkaline Earth Metals (A-Level Chemistry) Group 2, The Alkaline Earth Metals Introduction to Group 2 Elements Group 2, The Alkaline Earth Metals
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
